 The unit that provides this link is the mole (mol). To analyze the transformations that occur between individual atoms or molecules in a chemical reaction it is therefore absolutely essential for chemists to know how many atoms or molecules are contained in a measurable quantity in the laboratory-a given mass of sample. In the laboratory, for example, the masses of compounds and elements used by chemists typically range from milligrams to grams, while in industry, chemicals are bought and sold in kilograms and tons. Because the masses of individual atoms are so minuscule (on the order of 10 −23 g/atom), chemists do not measure the mass of individual atoms or molecules. The problem for Dalton and other early chemists was to discover the quantitative relationship between the number of atoms in a chemical substance and its mass. We also described the law of multiple proportions, which states that the ratios of the masses of elements that form a series of compounds are small whole numbers. In Dalton’s theory each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. The same calculation can also be done in a tabular format, which is especially helpful for more complex molecules: The number of grams of KClO3 will be 306.\right ) \right ] \) Multiply the given number of moles (2.50 mol) by the molar mass (122.548 g/mol) to get the grams.The molar mass of KClO3 is 122.548 g/mol. Problem: Convert 2.50 moles of KClO3 to grams. 
The number of moles of KMnO4 will be 0.158Īnd here is how you should enter this problem into the calculator above: grams to moles problem solution Moles to grams example problem Divide the given mass (25.0 g) by the molar mass (158.032 g/mol) to get the moles.The molar mass of KMnO4 is 158.032 g/mol. Problem: Convert 25.0 grams of KMnO4 to moles.įind out the molar mass of the substance (hint: you can use Molar mass of the substance alone to calculate molar mass). 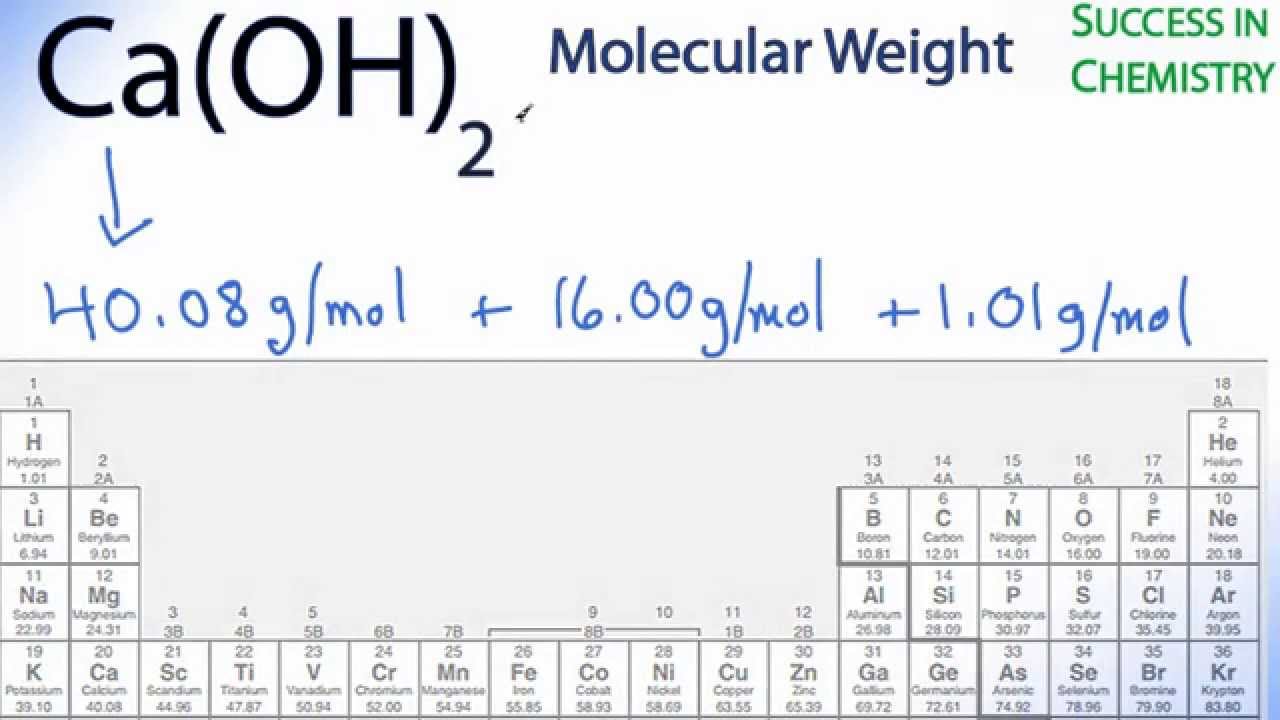
Multiplying by the molar mass constant ensures that the calculation is dimensionally correct: standard relative atomic masses are dimensionless quantities (i.e., pure numbers), whereas molar masses have units (in this case, grams/mole).Īnd this is where our grams to moles/moles to grams calculator shines, thanks to our other calculator - Molar mass of the substance, which calculates molar mass for a substance given the formula.Īll you need to do is correctly enter your formula, choose whether you want a conversion from grams to moles or a conversion from moles to grams, and, in case of g to mol, enter the mass, or, in case of mol to g, enter the moles. The molar mass of a compound is given by the sum of the standard atomic weight (namely, the standard relative atomic mass) of the atoms which form the compound multiplied by the molar mass constant. The molar mass of atoms of an element is given by the standard relative atomic mass of the element multiplied by the molar mass constant, 1 × 10−3 kg/mol = 1 g/mol. The molar mass is a physical property defined as the mass of a given substance (chemical element or chemical compound) divided by the amount of substance. You need to multiply the molar mass of the substance by the number of moles:Īs you can see, the most difficult task here is finding out the molar mass of the substance. 
You need to divide the mass of the substance by the molar mass of the substance: molar mass of the substance in grams/mole How to convert grams to moles? There is a simple relation between grams and moles: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
